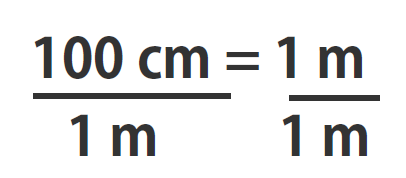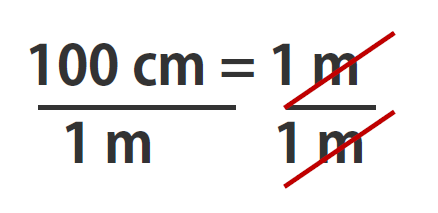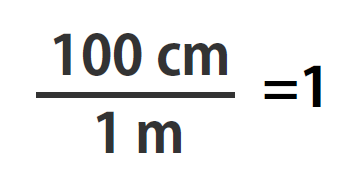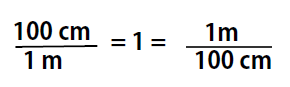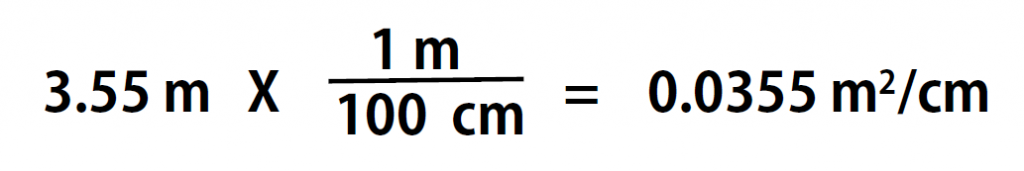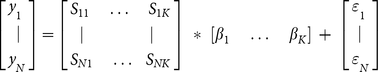Home » Student Resources » Online Chemistry Textbooks » CH104: Chemistry and the Environment » CH104 – Chapter 1: Measurements in Chemistry
MenuCH104: Chemistry and the Environment
Chapter 1 – Measurements in Chemistry
This content can also be downloaded as an printable PDF or an interactive PDF. For the interactive PDF, adobe reader is required for full functionality.
This text is published under creative commons licensing, for referencing and adaptation, please click here.
Sections:
Section 1: Chemistry and Matter
What is Chemistry?
Physical and Chemical Properties
Elements and Compounds
Mixtures
States of Matter
Section 2: How Scientists Study Chemistry
The Scientific Method
Section 3: Scientific Notation
Video Tutorial
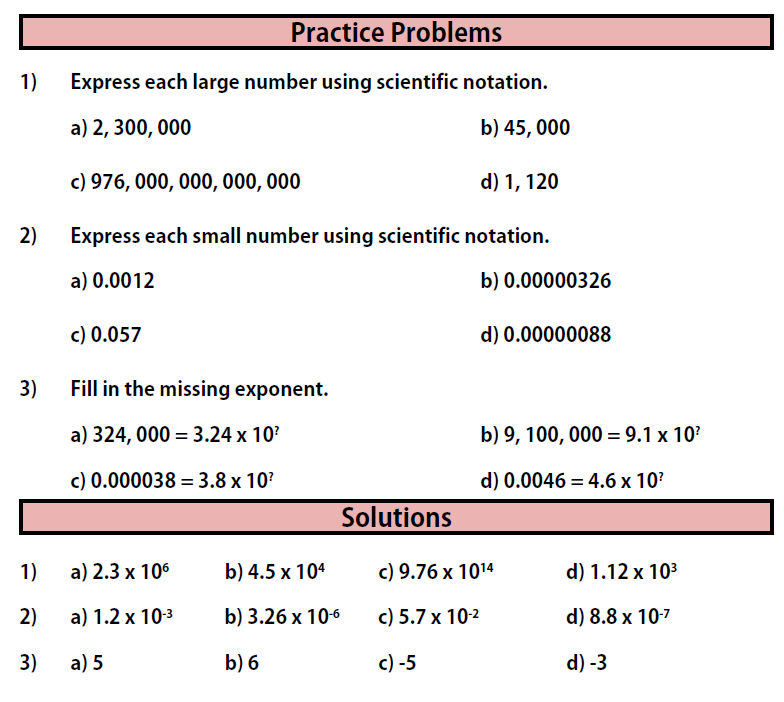
Practice Problems
Section 4: Units of Measurement
International System of Units and the Metric System
Derived SI Units
Section 5: Making Measurements in the Lab
Precision vs. Accuracy
Significant Figures
Exact Numbers
Rules of Rounding
Video Tutorial
Calculations with Significant Figures
Conversions and the Importance of Units
Conversion Factors
Section 6: Focus on the Environment – Plastics and Ocean Pollution
Section 7: Chapter Summary
Section 8: Chapter Homework
Section 9: References
Section 1: Chemistry and Matter
What is Chemistry?
Everything around us is made up of chemicals. From the color that makes a rose so red to the gasoline that fills our cars and the silicon chips that power our computers and cell phones…Chemistry is everywhere! Understanding how chemical molecules form and interact to create complex structures enables us to harness the power of chemistry and use it, just like a toolbox, to create many of the modern advances that we see today. This includes advances in medicine, communication, transportation, building infrastructure, food science and agriculture, and nearly every other technical field that you can imagine.
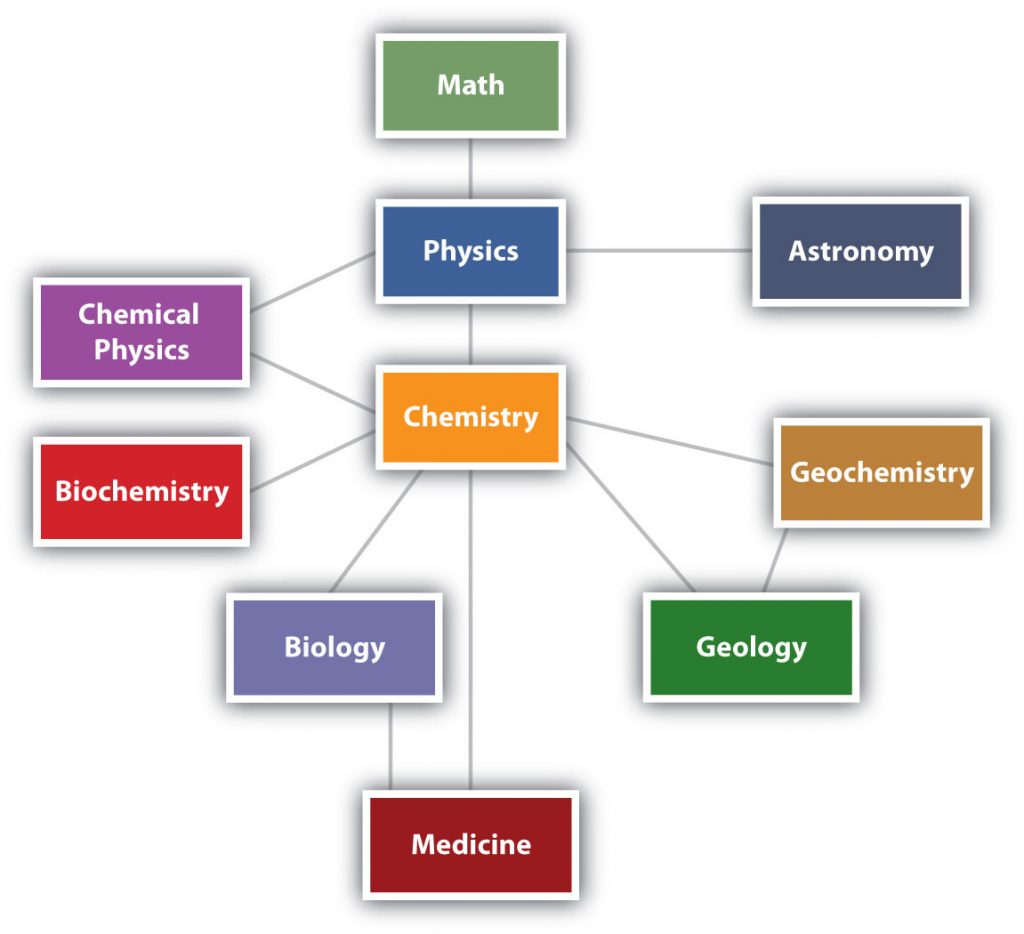
Chemistry is one branch of science. Science is the process by which we learn about the natural universe by observing, testing, and then generating models that explain our observations. is the process by which we learn about the natural universe by observing, testing, and then generating models that explain our observations. Because the physical universe is so vast, there are many different branches of science (Figure 1.1). Thus, chemistry is the study of matter, biology is the study of living things, and geology is the study of rocks and the earth. Mathematics is the language of science, and we will use it to communicate some of the ideas of chemistry.
Although we divide science into different fields, there is much overlap among them. For example, some biologists and chemists work in both fields so much that their work is called biochemistry. Similarly, geology and chemistry overlap in the field called geochemistry. Figure 1.1 shows how many of the individual fields of science are related.
Figure 1.1: The Relationships Between Some of the Major Branches of Science. Chemistry lies more or less in the middle, which emphasizes its importance to many branches of science.
Physical vs. Chemical Properties
Part of understanding matter is being able to describe it. One way chemists describe matter is to assign different kinds of properties to different categories. The properties that chemists use to describe matter fall into two general categories. Physical properties are characteristics that describes matter, such as boiling point, melting point and color. Physical Changes, such as melting a solid into a liquid, do not alter the chemical structure of that matter. Chemical properties are characteristics that describe how the chemical structure of matter changes during a chemical reaction. An example of a chemical property is flammability—a materials ability to burn—because burning (also known as combustion) changes the chemical composition of a material.
Elements and Compounds
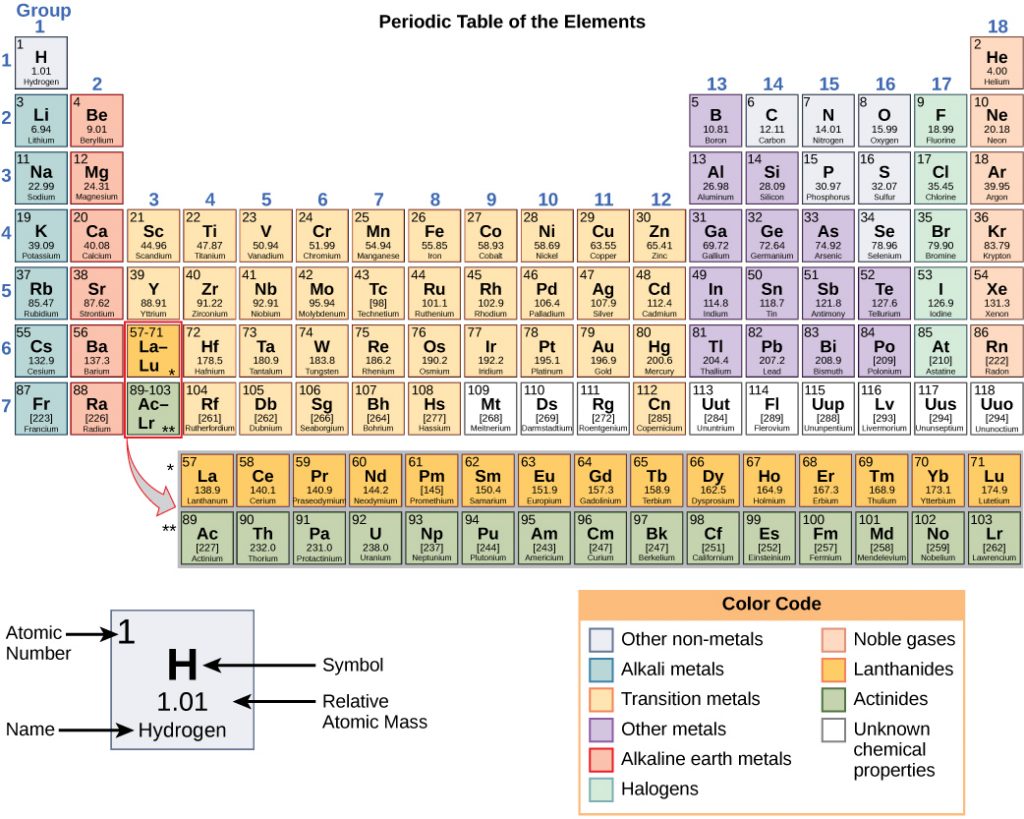
Any sample of matter that has the same physical and chemical properties throughout the sample is called a substance. There are two types of substances. A substance that cannot be broken down into chemically simpler components is an element. Aluminum, which is used in soda cans, is an element. A substance that can be broken down into chemically simpler components (because it has more than one element) is a compound. Water is a compound composed of the elements hydrogen and oxygen. Today, there are about 118 elements in the known universe which are organized on a fundamental chart called the Periodic Table of Elements (Fig. 1.2). In contrast, scientists have identified tens of millions of different compounds to date.
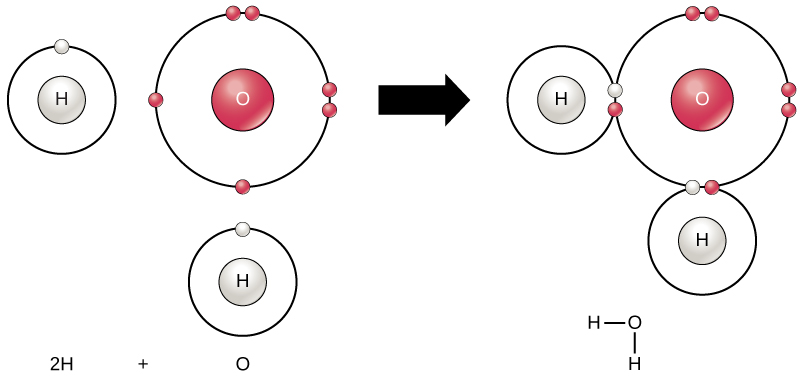
The smallest part of an element that maintains the identity of that element is called an atom. Atoms are extremely tiny; to make a line 1 inch long, you would need 217 million iron atoms! Similarly, the smallest part of a compound that maintains the identity of that compound is called a molecule. Molecules are composed of atoms that are attached together and behave as a unit (Fig. 1.2). Scientists usually work with millions of atoms and molecules at a time. When a scientist is working
Figure 1.2: (Upper Panel) The Periodic Table of the Elements is an organized chart that contains all of the known chemical elements. (Lower Panel) To the left of the arrow is shown one atom of oxygen and two atoms of hydrogen. Each of these represent single elements. When they are combined on the righthand side, they form a single molecule of water (H2O). Note that water is defined as a compound, because each single molecule is made up of more than one type of element, in this case, one atom of oxygen with two atoms of hydrogen.
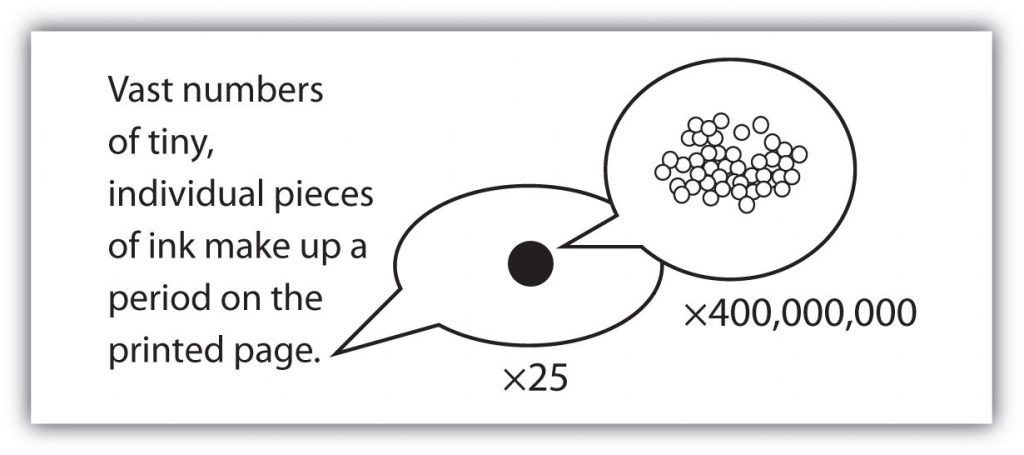
with large numbers of atoms or molecules at a time, the scientist is studying the macroscopic view of the universe. However, scientists can also describe chemical events on the level of individual atoms or molecules, which is referred to as the microscopic viewpoint. We will see examples of both macroscopic and microscopic viewpoints throughout this book (Figure 1.3).
Figure 1.3: How many molecules are needed for a period in a sentence? Although we do not notice it from a macroscopic perspective, matter is composed of microscopic particles so tiny that billions of them are needed to make a speck that we can see with the naked eye. The X25 and X400,000,000 indicate the number of times the image is magnified.
Mixtures
A material composed of two or more substances is a mixture. In a mixture, the individual substances maintain their chemical identities. Many mixtures are obvious combinations of two or more substances, such as a mixture of sand and water. Such mixtures are called heterogeneous mixtures. In some mixtures, the components are so intimately combined that they act like a single substance even though they are not. Mixtures with a consistent composition throughout are called homogeneous mixtures Homogeneous mixtures that are mixed so thoroughly that neither component can be observed independently of the other are called solutions. Sugar dissolved in water is an example of a solution. A metal alloy, such as steel, is an example of a solid solution. Air, a mixture of mainly nitrogen and oxygen, is a gaseous solution.
Figure 1.4: Heterogeneous vs. Homogeneous Mixtures. A mixture contains more than one substance. In the upper panel you see an example of a heterogeneous mixture of oil and water. The mixture is heterogeneous because you can visibly see two different components in the mixture. In the lower panel, you see an example of a homogeneous mixture, coffee. It is homogeneous because you cannot distinguish the many different components that make up a cup of coffee (water; caffeine; coffee alkaloids and tannins). It looks the same throughout. If the mixture is homogeneous and is also see through or clear, it is called a solution. In our example, the coffee is a solution; however, a concentrated espresso may be very opaque and would only be homogeneous mixture, not a solution.
States of Matter
Another way to classify matter is to describe it as a solid, a liquid, or a gas, which was done in the examples of solutions, above. These three descriptions, each implying that the matter has certain physical properties, represent the three phases of matter. A solid has a definite shape and a definite volume. Liquids have a definite volume but not a definite shape; they take the shape of their containers. Gases have neither a definite shape nor a definite volume, and they expand to fill their containers. We encounter matter in each phase every day. In fact, we regularly encounter water in all three phases: ice (solid), water (liquid), and steam (gas).
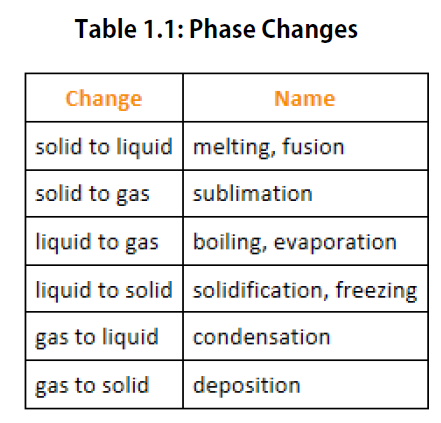
We know from our experience with water that substances can change from one phase to another if the conditions are right. Typically, varying the temperature of a substance (and, less commonly, the pressure exerted on it) can cause a phase change or a physical process in which a substance goes from one phase to another (Figure 1.5). Phase changes have particular names depending on what phases are involved, as summarized in Table 1.1.
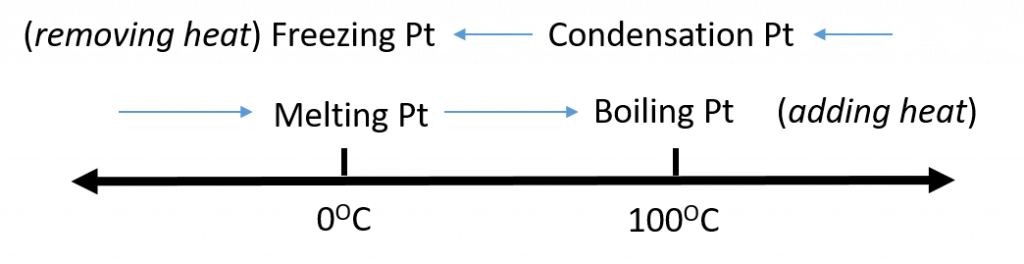
Figure 1.5. Analyzing Phase Changes. (Upper panel) A photo of boiling water demonstrates the phase change of water from the liquid to the gaseous phase. Note that phase changes are a physical property of a molecule. The water is still chemically the same (H2O) in the solid, liquid, or gaseous state. (Lower panel) Change in temperature can cause phase changes . Above is the temperature scale for the phase changes of water. If you add heat to solid ice, water will melt at 0oC and boil at 100oC. If you remove heat from gaseous water, it will condense into the liquid state at 100oC and freeze at 0oC.
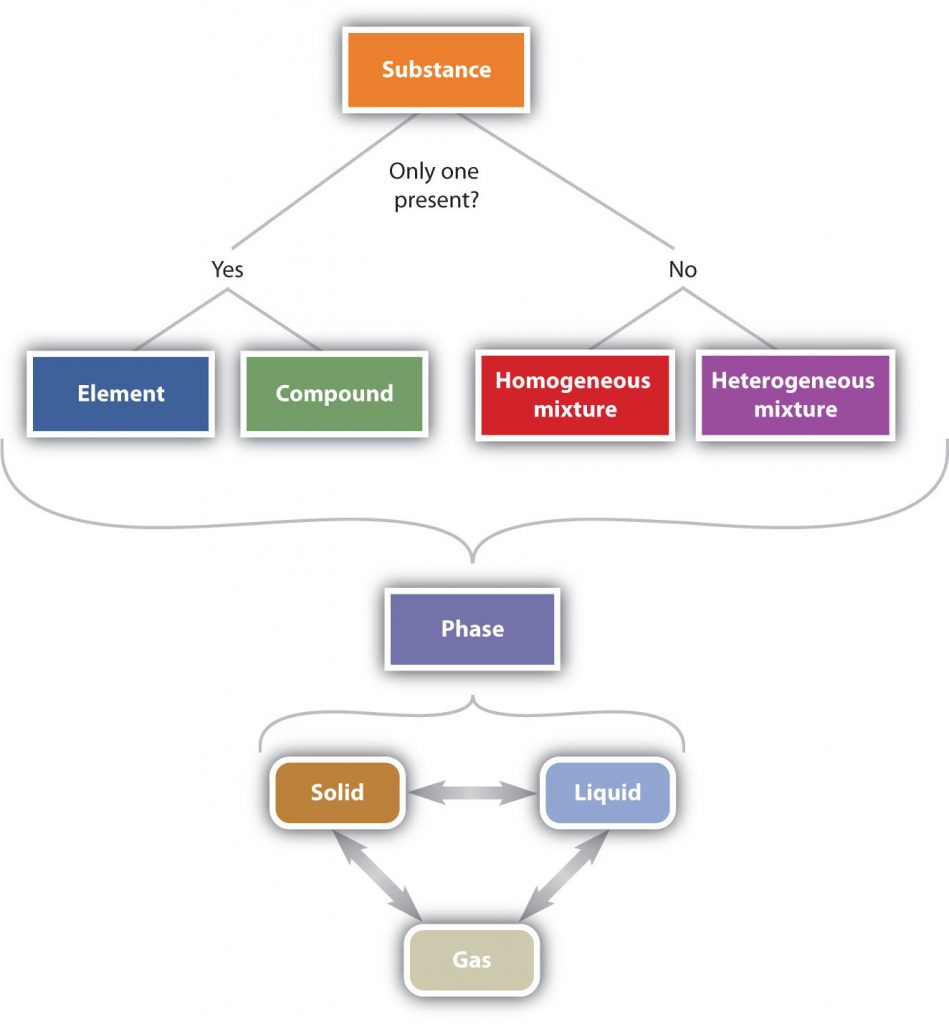
In summary, Figure 1.6 “The Classification of Matter” illustrates the relationships between the different ways matter can be classified.
Figure 1.6 The Classification of Matter. Matter can be classified in a variety of ways depending on its properties
Section 2: How Scientists Study Chemistry
The Scientific Method
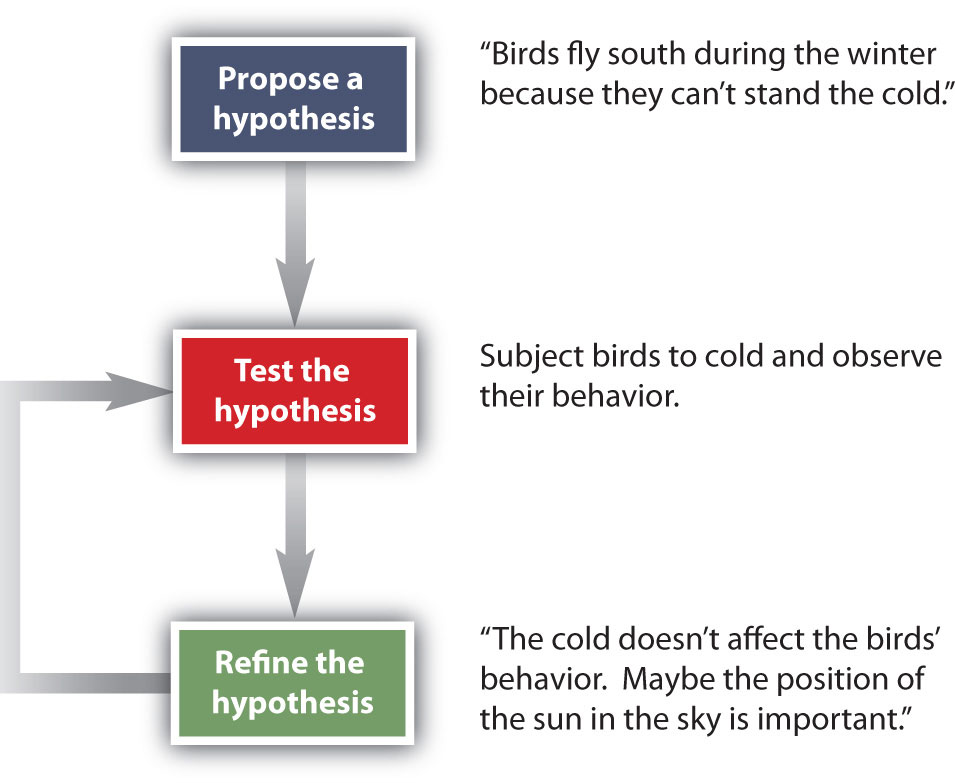
How do scientists work? Generally, they follow a process called the scientific method. The scientific method is an organized procedure for learning answers to questions. To find the answer to a question (for example, “Why do birds fly toward Earth’s equator during the cold months?”), a scientist goes through the following steps, which are also illustrated in Figure 1.7.
Figure 1.7 The General Steps of the Scientific Method. The steps may not be as clear-cut in real life as described here, but most scientific work follows this general outline.
Propose a hypothesis. A scientist generates a testable idea, or hypothesis, to try to answer a question or explain how the natural universe works. Some people use the word theory in place of hypothesis, but the word hypothesis is the proper word in science. For scientific applications, the word theory is a general statement that describes a large set of observations and data. A theory represents the highest level of scientific understanding, and is built from a wide array of factual knowledge or data.
Test the hypothesis. A scientist evaluates the hypothesis by devising and carrying out experiments to test it. If the hypothesis passes the test, it may be a proper answer to the question. If the hypothesis does not pass the test, it may not be a good answer.
Refine the hypothesis if necessary. Depending on the results of experiments, a scientist may want to modify the hypothesis and then test it again. Sometimes the results show the original hypothesis to be completely wrong, in which case a scientist will have to devise a new hypothesis.
Not all scientific investigations are simple enough to be separated into these three discrete steps. But these steps represent the general method by which scientists learn about our natural universe.
Section 3: Scientific Notation
The study of chemistry can involve numbers that are very large. It can also involve numbers that are very small. Writing out such numbers and using them in their long form is problematic, because we would spend far too much time writing zeroes, and we would probably make a lot of mistakes! There is a solution to this problem. It is called scientific notation.
Scientific notation allows us to express very large and very small numbers using powers of 10.
Recall that:
100 = 1 101 = 10 102 = 100
103 = 1000 104 = 10000 105 = 100000
As you can see, the power to which 10 is raised is equal to the number of zeroes that follow the 1. This will be helpful for determining which exponent to use when we express numbers using scientific notation.
Let us take a very large number:
579, 000, 000, 000
and express it using scientific notation.
First, we find the coefficient, which is a number between 1 and 10 that will be multiplied by 10 raised to some power.
Our coefficient is: 5.79
This number will be multiplied by 10 that is raised to some power. Now let us figure out what power that is.
We can do this by counting the number of positions that stand between the end of the original number and the new position of the decimal point in our coefficient.
5 . 7 9 0 0 0 0 0 0 0 0 0
↑ ↑
How many positions are there?
We can see that there are 11 positions between our decimal and the end of the original number. This means that our coefficient, 5.79, will be multiplied by 10 raised to the 11th power.
Our number expressed in scientific notation is:
5.79 x 1011
But what about very small numbers?
You may recall that:
10-1 = 0.1 10-2 = 0.01 10-3 = 0.001
10-4 = 0.0001 10-5 = 0.00001
The number of spaces to the right of the decimal point for our 1 is equal to the number in the exponent that is behind the negative sign. This is useful to keep in mind when we express very small numbers in scientific notation.
Here is a very small number:
0.0000642
Let us express this number using scientific notation.
Our coefficient will be 6.42
This number will be multiplied by 10 raised to some power, which will be negative. Let us figure out the correct power. We can figure this out by counting how many positions stand between the decimal point in our coefficient and the decimal point in our original number.
0 . 0 0 0 0 6 4 2
↑ ↑
How many positions?
There are 5 positions between our new decimal point and the decimal point in the original number, so our coefficient will be multiplied by 10 raised to the negative 5th power.
Our number written in scientific notation is:
6.42 x 10-5
You can use these methods to express any large or small number using scientific notation.
VIDEO TUTORIAL FOR SIGNIFICANT FIGURES:
Section 4: Units of Measurement
International System of Units and the Metric System
The International System of Units, abbreviated SI from the French Système International D’unités, is the main system of measurement units used in science. Since the 1960s, the International System of Units has been internationally agreed upon as the standard metric system. The SI base units are based on physical standards. The definitions of the SI base units have been and continue to be modified and new base units added as advancements in science are made. Each SI base unit except the kilogram is described by stable properties of the universe.
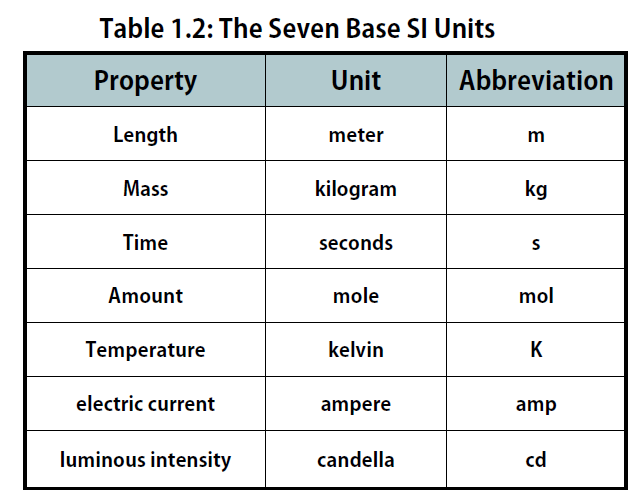
There are seven base units, which are listed in Table 1.2. Chemistry primarily uses five of the base units: the mole for amount, the kilogram for mass, the meter for length, the second for time, and the kelvin for temperature. The degree Celsius (oC) is also commonly used for temperature. The numerical relationship between kelvins and degrees Celsius is as follows
K = oC + 273
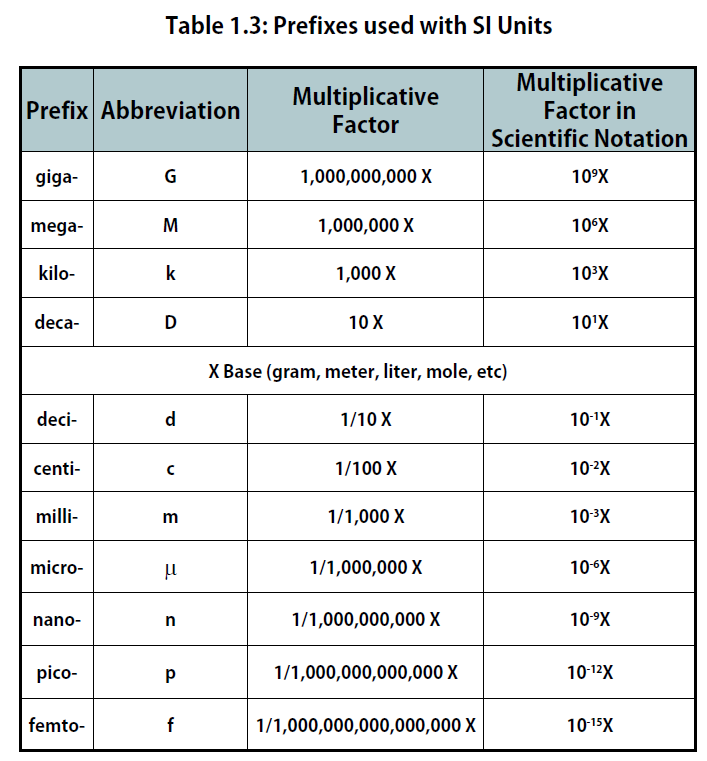
The size of each base unit is defined by international convention. For example, the kilogram is defined as the quantity of mass of a special metal cylinder kept in a vault in France (Figure 1.8). The other base units have similar definitions. The sizes of the base units are not always convenient for all measurements. For example, a meter is a rather large unit for describing the width of something as narrow as human hair. Instead of reporting the diameter of hair as 0.00012 m or even 1.2 × 10-4 m, SI also provides a series of prefixes that can be attached to the units, creating units that are larger or smaller by powers of 10, known as the metric system.
Figure 1.8 The Kilogram. The standard for the kilogram is a platinum-iridium cylinder kept in a speacial vault in France. Source: Wikimedea (https://commons.wikimedia.org/wiki/File:National_prototype_kilogram_K20_replica.jpg)
Common prefixes and their multiplicative factors are listed in Table 1.3 “Prefixes Used with SI Units”. (Perhaps you have already noticed that the base unit kilogram is a combination of a prefix, kilo- meaning 1,000 ×, and a unit of mass, the gram.) Some prefixes create a multiple of the original unit: 1 kilogram equals 1,000 grams (or 1 kg = 1,000 g), and 1 megameter equals 1,000,000 meters (or 1 Mm = 1,000,000 m). Other prefixes create a fraction of the original unit. Thus, 1 centimeter equals 1/100 of a meter, 1 millimeter equals 1/1,000 of a meter, 1 microgram equals 1/1,000,000 of a gram, and so forth.
The basic unit of mass in the International System of Units is the kilogram. A kilogram is equal to 1000 grams. A gram is a relatively small amount of mass and so larger masses are often expressed in kilograms. When very tiny amounts of matter are measured, we often use milligrams which are equal to 0.001 gram. There are numerous larger, smaller, and intermediate mass units that may also be appropriate. At the end of the 18th century, a kilogram was the mass of a liter of water. In 1889, a new international prototype of the kilogram was made of a platinum-iridium alloy. The kilogram is equal to the mass of this international prototype, which is held in Paris, France.
Mass and weight are not the same thing. Although we often use the terms mass and weight interchangeably, each one has a specific definition and usage. The mass of an object is a measure of the amount of matter in it. The mass (amount of matter) of an object remains the same regardless of where the object is placed. For example, moving a brick to the moon does not cause any matter in it to disappear or be removed.
The weight of an object is determined by the force that gravitation exerts upon the object. The weight is equal to the mass of the object times the local acceleration of gravity. Thus, on the Earth, weight is determined by the force of attraction between the object and the Earth. Since the force of gravity is not the same at every point on the Earth’s surface, the weight of an object is not constant. The gravitational pull on the object varies depending on where the object is with respect to the Earth or other gravity-producing object. For example, a man who weighs 180 pounds on Earth would weigh only 45 pounds if he were in a stationary position, 4,000 miles above the Earth’s surface. This same man would weigh only 30 pounds on the moon because the moon’s gravity is only one-sixth that of Earth. The mass of this man, however, would be the same in each situation. For scientific experiments, it is important to measure the mass of a substance rather than the weight to retain consistency in the results regardless of where you are performing the experiment.
Length
The SI unit of length is the meter. In 1889, the definition of the meter was a bar of platinum-iridium alloy stored under conditions specified by the International Bureau of Standards. In 1960, this definition of the standard meter was replaced by a definition based on a wavelength of krypton-86 radiation. In 1983, that definition was replaced by the following: the meter is the length of the path traveled by light in a vacuum during a time interval of a second.
Temperature
When used in a scientific context, the words heat and temperature do NOT mean the same thing. Temperature represents the average kinetic energy of the particles that make up a material. Increasing the temperature of a material increases its thermal energy. Thermal energy is the sum of the kinetic and potential energy in the particles that make up a material. Objects do not “contain” heat; rather they contain thermal energy. Heat is the movement of thermal energy from a warmer object to a cooler object. When thermal energy moves from one object to another, the temperature of both objects change.
A thermometer is a device that measures temperature. The name is made up of “thermo” which means heat and “meter” which means to measure. The temperature of a substance is directly proportional to the average kinetic energy it contains. In order for the average kinetic energy and temperature of a substance to be directly proportional, it is necessary that when the temperature is zero, the average kinetic energy must also be zero. It was necessary for use in calculations in science for a third temperature scale in which zero degrees corresponds with zero kinetic energy, that is, the point where molecules cease to move. This temperature scale was designed by Lord Kelvin. Lord Kelvin stated that there is no upper limit of how hot things can get, but there is a limit as to how cold things can get. In 1848, William Lord Kelvin developed the idea of absolute zero, which is the temperature at which molecules stop moving and therefore, have zero kinetic energy. This is known as the Kelvin temperature scale.
The Celcius scale is based on the freezing point and boiling point of water. Thus, 0oC is the freezing point of water, whereas 100oC is the boiling point of water. Most of us are familiar with temperatures that are below the freezing point of water. It should be apparent that even though the air temperature may be -5oC, the molecules of air are still moving (i.e. 0oC is not absolute zero). Substances like oxygen gas and nitrogen gas have already melted and boiled to vapor at temperatures below -150oC.
The Fahrenheit scale is also defined by the freezing point and boiling points of water. However, the scale is different from that of the Kelvin and Celsius scales. In the Fahrenheit scale, the freezing point of water is 32oF and the boiling point of water is 212oF. To convert between the Fahrenheit scale and the Celsius scale , the following conversions can be used:
[oC] = ([oF] -32) × 5/9 or [oF] = [oC] × 9/5 + 32
The Kelvin temperature scale has its zero at absolute zero (determined to be -273.15oC), and uses the same degree scale as the Celsius scale. Therefore, the mathematical relationship between the Celsius scale and the Kelvin scale is
K = oC + 273.15
In the case of the Kelvin scale, the degree sign is not used. Temperatures are expressed simply as 450 K, and are always positive.
Time
The SI unit for time is the second. The second was originally defined as a tiny fraction of the time required for the Earth to orbit the Sun. It has since been redefined several times. The definition of a second (established in 1967 and reaffirmed in 1997) is: the duration of 9,192,631,770 periods of the radiation corresponding to the transition between the two hyperfine levels of the ground state of the cesium-133 atom.
Amount
Chemists use the term mole to represent a large number of atoms or molecules. Just as a dozen implies 12 things, a mole (mol) represents 6.022 × 1023 things. The number 6.022 × 1023, called Avogadro’s number after the 19th-century chemist Amedeo Avogadro, is the number we use in chemistry to represent macroscopic amounts of atoms and molecules. Thus, if we have 6.022 × 1023 Oxygen atoms, we say we have 1 mol of Oxygen atoms. If we have 2 mol of Na atoms, we have 2 × (6.022 × 1023) Na atoms, or 1.2044 × 1024 Na atoms. Similarly, if we have 0.5 mol of benzene (C6H6) molecules, we have 0.5 × (6.022 × 1023) C6H6 molecules, or 3.011 × 1023 C6H6 molecules.
Derived units are combinations of SI base units. Units can be multiplied and divided, just as numbers can be multiplied and divided. For example, the area of a square having a side of 2 cm is 2 cm × 2 cm, or 4 cm2 (read as “four centimeters squared” or “four square centimeters”). Notice that we have squared a length unit, the centimeter, to get a derived unit for area, the square centimeter.
Volume
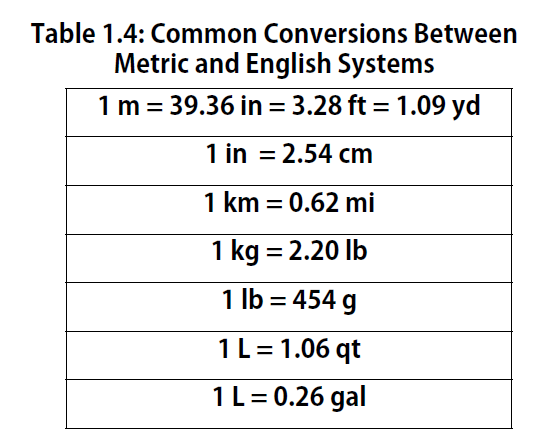
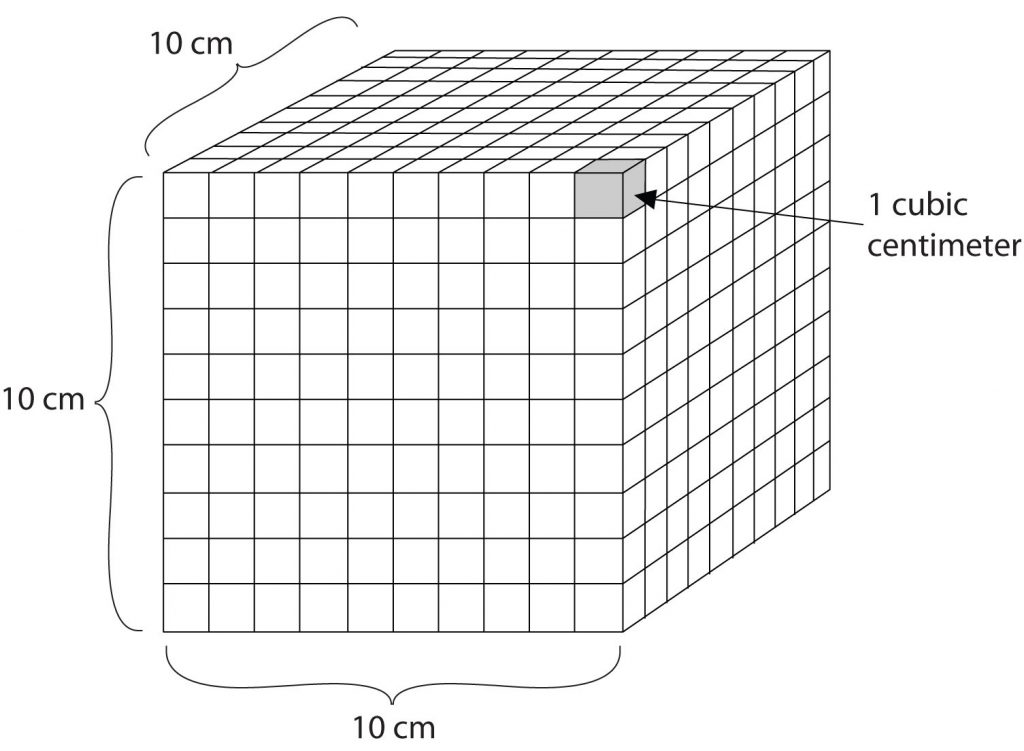
Volume is an important quantity that uses a derived unit. Volume is the amount of space that a given substance occupies and is defined geometrically as length × width × height. Each distance can be expressed using the meter unit, so volume has the derived unit m × m × m, or m3 (read as “meters cubed” or “cubic meters”). A cubic meter is a rather large volume, so scientists typically express volumes in terms of 1/1,000 of a cubic meter. This unit has its own name—the liter (L). A liter is a little larger than 1 US quart in volume. (Table 1.4) gives approximate equivalents for some of the units used in chemistry.) As shown in Figure 1.9 “The Liter”, a liter is also 1,000 cm3. By definition, there are 1,000 mL in 1 L, so 1 milliliter and 1 cubic centimeter represent the same volume.
1 mL = 1 cm3
Figure 1.9: The Liter. A liter is defined as a cube that is 10 cm (1/10th of a meter) on a side. A milliliter, 1/1000th of a liter, is equal to 1 cubic centimeter (1 cm3).
Energy
Energy, another important quantity in chemistry, is the ability to perform work. Moving a box of books from one side of a room to the other side, for example, requires energy. It has a derived unit of kg·m2/s2. (The dot between the kg and m2 units implies the units are multiplied together and then the whole term is divided by s2.) Because this combination is cumbersome, this collection of units is redefined as a joule (J), which is the SI unit of energy. An older unit of energy, the calorie (cal), is also widely used. There are:
4.184 J = 1 cal
Note that this differs from our common use of the big ‘Calorie’or ‘Cal’ listed on food packages in the United States. The big ‘Cal’ is actually a kilocalorie or kcal (Fig 1.10) Note that all chemical processes or reactions occur with a simultaneous change in energy and that energy can be stored in chemical bonds.
Figure 1.10: The Difference between kilocalories in Scientific and Common Use. Calories represented on food packaging actually refer to kilocalories in scientific terms.
Density
Density is defined as the mass of an object divided by its volume; it describes the amount of matter contained in a given amount of space.
density=mass/volume
Thus, the units of density are the units of mass divided by the units of volume: g/cm3 or g/mL (for solids and liquids, respectively), g/L (for gases), kg/m3, and so forth. For example, the density of water is about 1.00 g/mL, while the density of mercury is 13.6 g/mL. Mercury is over 13 times as dense as water, meaning that it contains over 13 times the amount of matter in the same amount of space. The density of air at room temperature is about 1.3 g/L.
Section 5: Making Measurements in the Lab
Precision vs. Accuracy
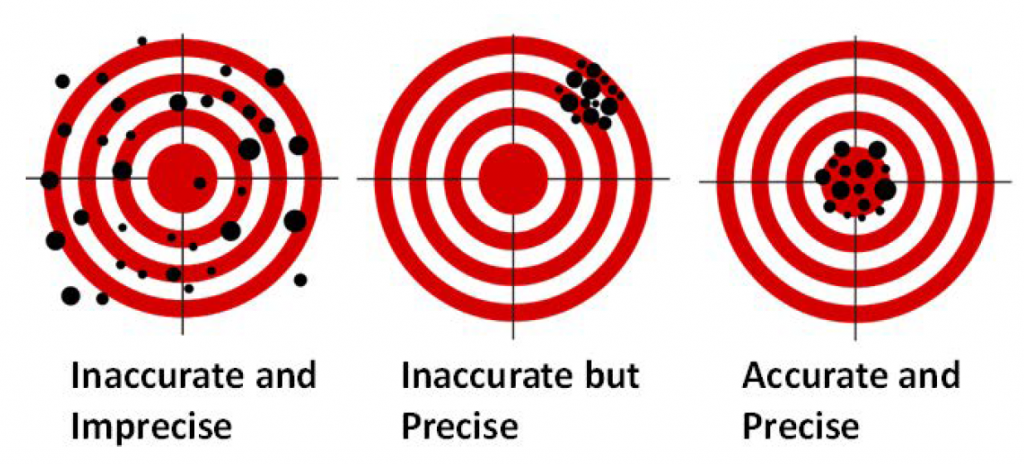
It is important to note the different terminology we use when talking in science. One such set of terminology is precision and accuracy. Although precision and accuracy are often used interchangeably in the non-scientific community, the difference between the terms is extremely important to realize. Precision tells you how close two measurements are to one another, while accuracy tells you how close a measurement is to the known value. A measurement can be precise while not being accurate, or accurate but not precise; the two terms are NOT related. A good analogy can be found in a game of darts (Fig. 1.11). A player who always hits the same spot just to the left of the dart board would be precise but not very accurate. However, a dart player who is all over the board but hits the center of the board on average would be accurate but not precise. A good darts player, just like a good scientist, wants to be both precise and accurate.
Figure 1.11: Difference Between Accuracy and Precision. A game of darts can be used to show the difference between accuracy and precision.
Adapted from: https://upload.wikimedia.org/wikipedia/commons/thumb/5/5d/Reliability_and_validity.svg/717px-Reliability_and_validity.svg.png
Typically within the laboratory, accuracy is a measure of how well your equipment is calibrated. For example, if your balance is not calibrated correctly, you can make very precise, repeated measurements, but the measurements will not represent the true value. Precision, on the otherhand, is usually determined by how careful the scientist is in making measurements. If you are careless and spill part of your sample on the way, your measurements in repeated experiments will not be precise even if your balance is accurate.
Significant Figures
It is important to realize that values in scientific measurements are never 100% accurate. Our instruments only measure to a certain level of accuracy. Thus, we can pick different instruments to make a measurement based upon the level of accuracy we need for the experiment. Due to the inherent inaccuracy in any measured number we must keep track of the different levels of accuracy each number has with significant figures. Significant figures of a measured quantity are defined as all the digits known with certainty and the first uncertain, or estimated, digit. It makes no sense to report any digits after the first uncertain one, so it is the last digit reported in a measurement. Zeros are used when needed to place the significant figures in their correct positions. Thus, zeros may or may not be significant figures. Significant figures apply in the real world, as they allow us to quantify the accuracy of any type of measurement. To identify how many numbers in a measurement have significance, you can follow a discreet set of rules, shown below and to the right.
Figure 1.12: Measuring an Object to the Correct Number of Significant Figures.
How many digits should be shown in this measurement?
The correct answer is 3! The two that you know for sure + the estimated position…for this reading it would be close to 1.37
Exact Numbers
Exact numbers are numbers that are not measured by a scientific instrument. They are either used as definitions to define a concept or terminology, or they are made by counting the total of something present. An example of an exact number, would be the number of eggs in a carton or a defined unit such as there are 100 cm in 1 m. Exact numbers, such as the number of people in a room, DO NOT affect the number of significant figures in calculations made with measured values.
Rules of Rounding
In scientific operations, the rules of rounding may be a little bit different than the ones you are used to using. Normal rounding rules suggest that if a number is 4 or below, it should be rounded down to the lower number, whereas if it is 5 or higher, it should be rounded up. However, note that 5 is right in the middle and causes a problem when using these conventional rounding rules. If you have a large dataset of numbers that you need to round, using this rounding rule will lead to bias in your dataset (i.e. 4/9th of the time you will be rounding down, and 5/9th of the time you will be rounding up). In a large dataset, this bias is unacceptable.
In Scientific Rounding, we typically use a rule called ‘Rounding to the Even.’ In this rounding system the rules are the same for 4 and below, you round down to the lower number, and for 6 and above you round up to the higher number. However, if the number you are rounding is 5, then you round to the even number. This helps to alleviate the sample bias that can occur when rounding large datasets.
Example:
Round 26.65 to three significant figures.
Solution:
When you look at the 4th digit, it is a 5. So now you must look at the 3rd digit. In this case it is even, so you would round to the even and the final answer would be 26.6
Calculations with Significant Figures
The first thing to realize before performing any calculations in science is that all measured numbers are only as good as the instrument used to measure them. Even with the best instrument available the measured number will never be 100% exact. Scientists use the “good enough” rule of precision, meaning that we accept an inherent amount of imprecision from every measurement we take as long as the final result is close enough to where we want it to be. This concept becomes dangerous when we begin to use these “good enough” numbers for any calculations, if we aren’t careful to keep track of our significant figures our numbers can quickly lose their “good enough” status. To protect their “good enough” numbers, the scientific community has set forth certain rules for performing any calculations; in this section we need only concern ourselves with two very important rules: the Addition/Subtraction rule, and the Multiplication/Division rule.
Addition/Subtraction Rule:
- Find the number with the least number of decimals and keep track of the number of decimal places
- Perform the addition/subtraction
- Round the final answer to the least number of decimals found in Step 1
Multiplication/Division Rule:
- Count the number of significant figures in each number (keep track of the number of significant figures)
- Perform the multiplication/division
- Round your final answer to the lowest number of significant figures found in step 1
Calculating Complicated Problems:
- Using the order of operations, break the problem up into multiple steps
- Perform any addition/subtraction steps following the Addition/Subtraction rule (Do not round yet, just keep track of the correct number of decimals when finding the number of significant figures)
- Perform multiplication/division using the Multiplication/Division rule
- Round the final answer to the correct number of significant figures
Conversions and the Importance of Units
The ability to convert from one unit to another is an important skill. For example, a nurse with 50 mg aspirin tablets who must administer 0.2 g of aspirin to a patient, needs to know that 0.2 g equals 200 mg, so that 4 tablets are needed. Fortunately, there is a simple way to convert from one unit to another.
If you learned the SI units and prefixes described in Section 1.4 Units of Measurement”, then you know that 1 cm is 1/100th of a meter or:
100 cm = 1 m
Suppose we divide both sides of the equation by 1 m (both the number and the unit; Note that it is critically important to always write out your units! This avoids confusion and mistakes when making conversions.):
As long as we perform the same operation on both sides of the equals sign, the expression remains an equality. Look at the right side of the equation; it now has the same quantity in the numerator (the top) as it has in the denominator (the bottom). Any fraction that has the same quantity in the numerator and the denominator has a value of 1:
We know that 100 cm is 1 m, so we have the same quantity on the top and the bottom of our fraction, although it is expressed in different units. A fraction that has equivalent quantities in the numerator and the denominator but expressed in different units is called a conversion factor
Note that conversion factors can be written with either term in the numerator or denominator, and used as appropriate for the problem that you want to solve. This is because, both terms are equal to 1
Here is a simple example. How many centimeters are there in 3.55 m? Perhaps you can determine the answer in your head. If there are 100 cm in every meter, then 3.55 m equals 355 cm. To solve the problem more formally with a conversion factor, we first write the quantity we are given, 3.55 m. Then we multiply this quantity by a conversion factor, which is the same as multiplying it by 1. We can write 1 as 100cm/1m and multiply:
Because m, the abbreviation for meters, occurs in both the numerator and the denominator of our expression, they cancel out. The final step is to perform the calculation that remains once the units have been canceled. Note that it is CRITICAL to retain the right units in the final answer or it will not make sense. A generalized description of this process is as follows:
quantity (old units) × conversion factor = quantity (new units)
You may be wondering why we use a seemingly complicated procedure for a straightforward conversion. In later studies, the conversion problems you will encounter will not always be so simple. If you can master the technique of applying conversion factors, you will be able to solve a large variety of problems.
In the previous example, we used the fraction 100 cm/1 m as a conversion factor. Does the conversion factor 1 m/100 cm also equal 1? Yes, it does; it has the same quantity in the numerator as in the denominator (except that they are flip-flopped). Why did we not use that conversion factor? If we had used the second conversion factor, the original unit would not have canceled, and the result would have been meaningless. Here is what we would have gotten:
INCORRECT USE OF CONVERSION FACTOR!!
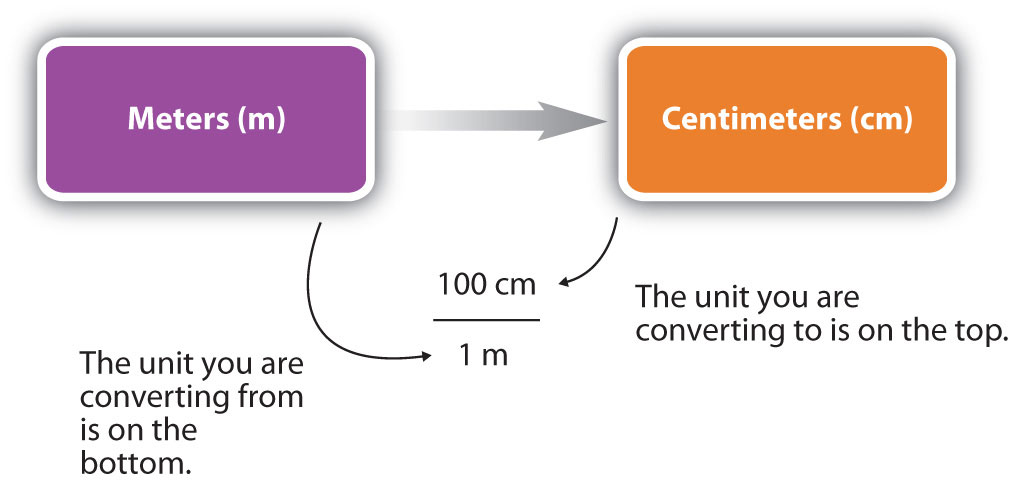
You can see that none of the units cancelled out. For the answer to be meaningful, we have to construct the conversion factor in a form that causes the original unit to cancel out. Figure 1.13 “A Concept Map for Conversions” shows a concept map for constructing a proper conversion.
Figure 1.13 A Concept Map for Conversions. This is how you construct a conversion factor to convert from one unit to another.
1.6 Focus on the Environment – Plastics and Ocean Pollution
In this section, we present a scientific study published by the Public Library of Science (PLOS) under the Creative Commons License. The original article can be accessed from the PLOS Open Access – Peer Reviewed Online Journal
The purpose of presenting this article is to give you experience with science in action. In section 1.2 you learned about the key components of the scientific method and experimental design. Two additional key elements that are critical for scientific studies are replicability and peer review. Replicability is defined as the ability of a scientific experiment or trial to be repeated to obtain a consistent result. This is a critical component of a scientific study related to the qualities of precision and accuracy discussed in section 1.5. Peer Review then offers a way for scientists to referee and evaluate the precision and accuracy of scientific studies before they can be published. During Scholary Peer Review the author’s scholarly work, research, or ideas are scrutinized by others who are experts in the same field before a paper describing this work is published within a journal or book.
The typical sections of a scientific article include the Abstract where the authors summarize the work and their findings, the Introduction where authors describe goal of the research and give background information, the Methods where authors describe in detail how the research was conducted (note that the methods should be given in enough detail to allow other research groups to repeat the work), The Results that presents the research data that was collected (usually in the form of figures and tables) and the Conclusions or Discussion where authors describe the implications of their work in the larger context of science and their field of research. Sometimes when reading research articles it is helpful to read the Abstract, Introduction and Discussion first to acquaint yourself with the purpose of the research and then focus on the Methods and Results to see if the research presented supports the authors conclusions and broader perspectives. Note that some articles also contain supplemental figures that support the article, but are not officially included as part of the article. They can appear with the main article or they may be published at another location and available for download. This article does contain supplemental figures at the end of the discussion section. In addition, peer reviewed science articles also contain a References section that provided the location of all source materials that was used to develop the written document.
The research article below focuses on the accumulation of floating plastic debris with the world’s oceans. Plastics represent a major pollution problem within our oceans, with the Center for Biological Diversity estimating that plastic floating debris covers approximately 40% of the worlds ocean surfaces. It represents a hazard to birds, fish, sea turtles and marine mammals that mistakenly ingest plastic particles as food or become entangled in plastic debris and drown. Plastics can also release and accumulate toxic substances, such as lead, cadmium, mercury, bisphenol-A, and pthalates, that have been linked with cancer, birth defects, immune system problems, and childhood developmental issues. In the article by Erickson, et al below, researchers are trying to establish a good estimate for how much plastic is currently floating in the ocean.
Figure 1.14 The Effects of Plastic Pollution in the World’s Oceans. Upper left panel: Mountain of Plastic. Photo credit: Top News, “Scientists Baffled by Mystery of Missing Ocean Plastic”, Jamie Williamson, http://www.topnews.in/usa/scientists-baffled-mystery-missing-ocean-plastic-25250. Upper right panel: Location of Plastic Islands in Ocean. Photo credit: Earthly Issues, Charles Welch, http://www.earthlyissues.com/plastic.htm Lower panel: Bird filled with plastic. Photo courtesy of http://www.simple-green-living.com, Simple Green Living, “One Step at a Time”
Homework Assignment: Please download the following CH104 Chapter 1 Environmental Assignment and as you read through the following research article complete the assignment. This is a nice assignment to have a class discussion about the scientific method.
Abstract
Plastic pollution is ubiquitous throughout the marine environment, yet estimates of the global abundance and weight of floating plastics have lacked data, particularly from the Southern Hemisphere and remote regions. Here we report an estimate of the total number of plastic particles and their weight floating in the world’s oceans from 24 expeditions (2007–2013) across all five sub-tropical gyres, costal Australia, Bay of Bengal and the Mediterranean Sea conducting surface net tows (N = 680) and visual survey transects of large plastic debris (N = 891). Using an oceanographic model of floating debris dispersal calibrated by our data, and correcting for wind-driven vertical mixing, we estimate a minimum of 5.25 trillion particles weighing 268,940 tons. When comparing between four size classes, two microplastic <4.75 mm and meso- and macroplastic >4.75 mm, a tremendous loss of microplastics is observed from the sea surface compared to expected rates of fragmentation, suggesting there are mechanisms at play that remove <4.75 mm plastic particles from the ocean surface.
Introduction
Plastic pollution is globally distributed across all oceans due to its properties of buoyancy and durability, and the sorption of toxicants to plastic while traveling through the environment [1], [2], have led some researchers to claim that synthetic polymers in the ocean should be regarded as hazardous waste [3]. Through photodegradation and other weathering processes, plastics fragment and disperse in the ocean [4], [5], converging in the subtropical gyres [6]–[9]. Generation and accumulation of plastic pollution also occurs in closed bays, gulfs and seas surrounded by densely populated coastlines and watersheds [10]–[13]
The impact of plastic pollution through ingestion and entanglement of marine fauna, ranging from zooplankton to cetaceans, seabirds and marine reptiles, are well documented [14]. Adsorption of persistent organic pollutants onto plastic and their transfer into the tissues and organs through ingestion [15] is impacting marine megafauna [16] as well as lower trophic-level organisms [17], [18] and their predators [19], [20]. These impacts are further exacerbated by the persistence of floating plastics, ranging from resin pellets to large derelict nets, docks and boats that float across oceans and transport microbial communities [21], algae, invertebrates, and fish [22] to non-native regions [23], providing further rationale to monitor (and take steps to mitigate) the global distribution and abundance of plastic pollution.
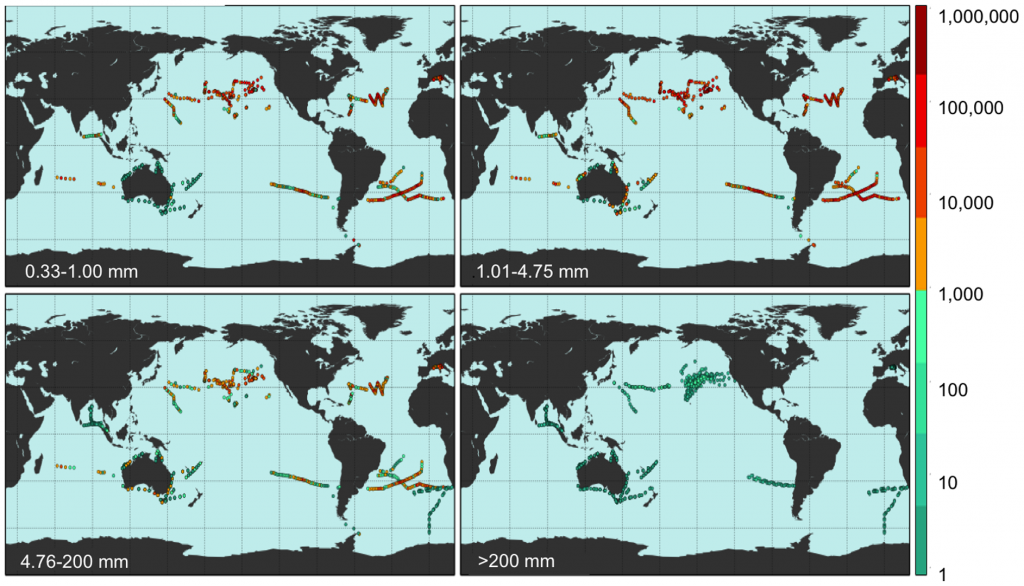
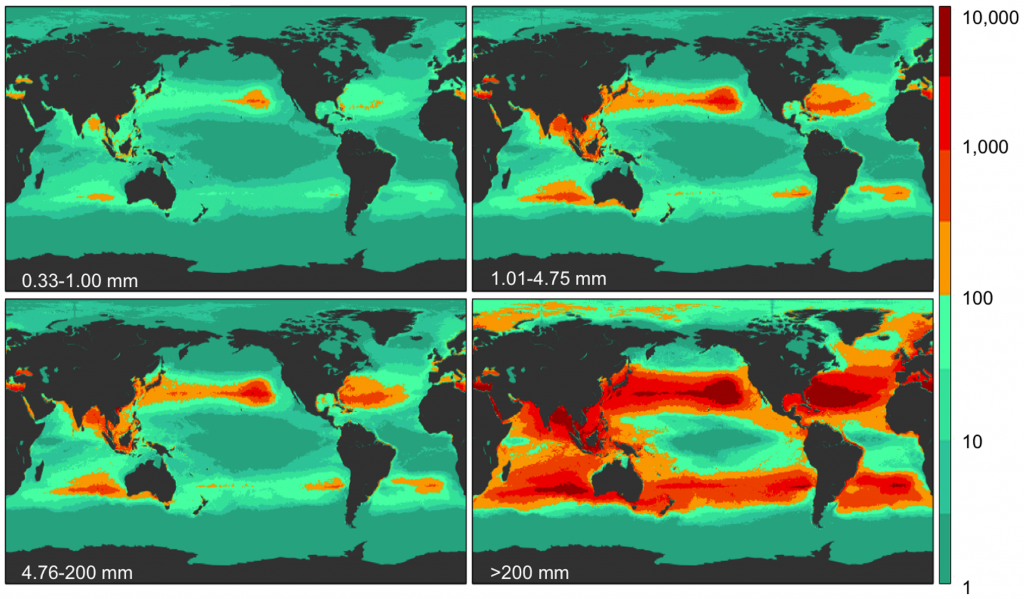
Despite oceanographic model predictions of where debris might converge [24] estimates of regional and global abundance and weight of floating plastics have been limited to microplastics <5 mm [19], [25]. Using extensive published and new data, particularly from the Southern Hemisphere subtropical gyres and marine areas adjacent to populated regions [7], [10], [13], [26], corrected for wind-driven vertical mixing [27], we populated an oceanographic model of debris distribution [28] to estimate global distribution and count and weight densities of plastic pollution in all sampled size classes. The oceanographic model assumes that amounts of plastic entering the ocean depend on three principal variables: watershed outfalls, population density and maritime activity. The dataset used in this model is based on expeditions from 2007–2013 (Table S1), surveying all five sub-tropical gyres (North Pacific, North Atlantic, South Pacific, South Atlantic, Indian Ocean) and extensive coastal regions and enclosed seas (Bay of Bengal, Australian coasts and the Mediterranean Sea), and include surface net tows (N = 680) and visual survey transects for large plastic debris (N = 891) totaling 1571 locations in all oceans (Fig 1). We also compared plastic pollution levels between oceans and across four size classes: 0.33–1.00 mm (small microplastics), 1.01–4.75 mm (large microplastics), 4.76–200 mm (mesoplastic), and >200 mm (macroplastic) (Fig. 1).
Figure 1. Field locations where count density was measured. Count density (pieces km−2; see colorbar) of marine plastic debris measured at 1571 stations from 680 net tows and 891 visual survey transects for each of four plastic size classes (0.33–1.00 mm, 1.01–4.75 mm, 4.76–200 mm, and >200 mm).
Materials and Methods
Net tow sample collection and analysis
Net tows were conducted using neuston nets with a standard mesh size of 0.33 mm towed between 0.5 and 2 m s−1 at the sea surface for 15–60 minutes outside of the vessel’s wake to avoid downwelling of debris. Samples were preserved in 5% formalin. Using a dissecting microscope, microplastic was manually separated from natural debris, sorted through stacked Tyler sieves into three size classes [7], [10], [12], then counted individually and weighed together. During sample analysis the identity of smaller microplastics was confirmed with buoyancy and hardness tests. All items were counted and weighed to the nearest 0.01 mg. Using these data, trawl dimensions and distance traveled, count (pieces km−2) and weight (g km−2) densities were estimated. The slow tow speed and the washing of the net between the tows when needed provided sufficient confidence that any variation in sample collection efficiency due to the net size, difference in tow speed or tow time were negligible.
Visual survey protocol
Visual survey transects of large plastic debris were carried out during expeditions to the South Pacific, North Pacific, South Atlantic, Indian Ocean, and waters around Australia, as well as part of the NOAA Trans-Pacific Marine Debris Survey in the North Pacific. Dedicated observers viewed the ocean surface on one side of the vessel out to 20 meters noting large debris items during timed observation periods [11], [13], [26], with start and stop positions used to calculate the area surveyed. Debris observations were broken into nine categories, four categories for fishing-related debris: buoy, line, net, and other fishing gear, and five categories for other plastics: bucket, bottle, foamed polystyrene, bag/film, or miscelaneous plastics (Table S2). Because observed debris cannot be collected and weighed, similar debris items in similar categories were collected from shorelines in northern-central Chile, South Africa, Atlantic coast of North America and the Hawaiian Archipelago to determine mean weights of items in the nine categories (Table S3). The two categories labeled ‘other fishing gear’ and ‘miscellaneous plastics’ were assigned a very conservative weight of 10 g per item. These mean weights were applied to visual survey transects to determine weight densities.
Description of the model
Particle tracking is accomplished in two stages, first a hydrodynamic model describes oceanic circulation and second virtual particles are introduced into the flow field and allowed to move freely through hydrodynamic forcing. For this study, ocean surface currents are extracted from the oceanic circulation modeling system HYCOM/NCODA [29]. The HYCOM model is forced by the US Navy’s Operational Global Atmospheric Prediction System (NOGAPS) and includes wind stress, wind speed, heat flux, and precipitation. The model provides systematic archiving of daily ocean circulation on a global scale with output data archived back to mid-2003. While the full HYCOM model contains 32 vertical layers, we only consider velocities in the surface layer as the principal driver of floating particles.
Velocity data extracted from HYCOM are then coupled to the Lagrangian particle-tracking model Pol3DD, which drives the dispersion of floating material. Pol3DD tracks and stores the origin, age, and trajectory information of individual particles [30]. Since wind driven currents are already expressed in the HYCOM hydrodynamic data, no additional wind stress terms were applied to the motion of particles. This model assumes that debris particles are mostly submerged in the water and extra forcing on potentially emerged parts of the debris is neglected.
Model calibration using empirical data from 1571 locations
In this study we determined abundances and mass of microplastics starting at the lowest size of 0.33 mm, which is a commonly used lower limit for pelagic microplastics [31]. The prefixes micro, meso and macro in relation to plastic pollution are poorly defined. Generally accepted microplastic boundaries are based on typical neuston net mesh size (0.33 mm) and an upper boundary of approximately 5.0 mm [31]. We have used 4.75 mm as our upper boundary for microplastic because this is a size for standard sieves used for sample analysis in most of the expeditions contributing data to this manuscript. Mesoplastic has a lower limit of 4.75 mm, and no defined upper limit. In this current study we set the upper boundary of mesoplastic at 200 mm, which represents a typical plastic water bottle, chosen because of its ubiquity in the ocean. Macroplastic has no established lower boundary, though we set it at 200 mm, while the upper boundary is unlimited. There is a clear need for consistent measures in the field [31], and herein we followed a practical approach using commonly employed boundaries and logistic considerations (net and sieve sizes) in order to integrate an extensive dataset that covers the entire global ocean, including areas that have never been sampled before.
Of the 1571 field locations that contributed count data (Fig. 1), a total of 1333 stations also had weight data (Fig. S4). All these data were used to calibrate the numerical model prediction of plastic count and weight density [28]. For the comparison, we fit the model results to measured data by a linear system of equations of the form:
where yi is the logarithm of a measured value of plastic count density (pieces km−2) or weight density (g km−2) for each of the N number of samples. K is the number of model output cases with sij a dimensionless model solution at the location of sample yi. βk and εN are the computed weighting coefficients and the error terms for a particular dimensionless model solution sij. This method can be used to fit an arbitrary number of model output cases to any number of measured data points producing a weighting coefficient and error term for each case.
In the model we used a set of three model results (K = 3), corresponding to different input scenarios [28]: urban development within watersheds, coastal population and shipping traffic. Values of β and ε are determined for both the concentration distribution (pieces km−2) and the weight distribution (g km−2) of each of the four size classes based on the linear system of equations. To compare the model results directly to the measured data, the weighting coefficient βk computed above is used to scale the model output for each of the output scenarios.
Adjusting estimated weight and count due to vertical distribution
Wind-driven mixing of the surface layer will drive particles downward, which causes underestimations of plastic in the ocean if relying on surface sampling only. We used a vertical distribution equation from Kukulka et al. [27], relating the ratio of the true number of particles/measured number of particles with the frictional velocity of water (u*w = [t/rw]1/2, where t is the wind stress and rw is the density of water).
Our data from 680 net tows includes Beaufort Scale sea states, each with a wind speed range. Before using the vertical distribution equation, we transformed these data into wind stress values, by applying the Smith [32] coefficient for sea surface wind stress (N/m2) as a function of wind speed (m/s). These data were then used in the vertical distribution equation to adjust the total particle count of plastic for each station.
To estimate the increased mass due to vertical distribution, we attributed the same percentage increase in particle count to particle weight.
Estimating expected particle counts based on fragmentation of large particles
We use conservative estimates of fragmentation rates to show that the model results of particle count in each size class differ substantially from our expected particle counts. To estimate fragmentation rates, we assumed that all particles, including the largest ones had a thickness of 0.2 mm. This assumption is conservative, because it is well known that many larger items have a wall thickness substantially larger than this. We assumed smaller particle sizes for the largest size classes, while for the smallest size class (0.33 mm–1.00 mm) we assumed a conservative particle diameter of 0.8 mm – this is substantially larger than most microplastics collected at the sea surface. Thus, our fragmentation estimates are highly conservative because for the macroplastics that generate plastic fragments we consider lower initial mass than commonly found at sea, while for the microplastics in our fragmentation exercise we consider larger particles than typically found at sea. Fragmentation of one macroplastic item (200 mm diameter) into typical mesoplastic fragments (50 mm diameter) would result in 16 particles, fragmentation of one 50 mm diameter mesoplastic item into typical large microplastics (2 mm diameter) results in 625 particles, and fragmentation of one large microplastic item (2 mm diameter) into small microplastics with a diameter of 0.8 mm results in 6.25 particles.
We then used these ratios in a stepwise approach to estimate particle counts in each size class based on the model results of particle count in the next-higher size category. For example, in the North Pacific the modeled data show 0.33×1010 particles in the macroplastic size class. Using our estimated fragmentation ratio of 1∶16 between macro and mesoplastic, we expect 5.33×1010 particles in the mesoplastic size class for the entire North Pacific. These fragmentation ratios between size categories are utilized to estimate the expected particle count for large and small microplastic particles. This stepwise approach is simplistic, because it assumes that the system is close to equilibrium. We recognize that rates of new plastic entering the ocean are unknown, as well as outputs of plastic due to beaching, sinking and mechanisms of degradation, and use these fragmentation estimates as first crude intent to reveal the dynamics of floating plastics in the oceans.
Ethics Statement
During these sampling procedures, no permits were required as we only collected plankton samples, and those samples were collected in international waters.
Results
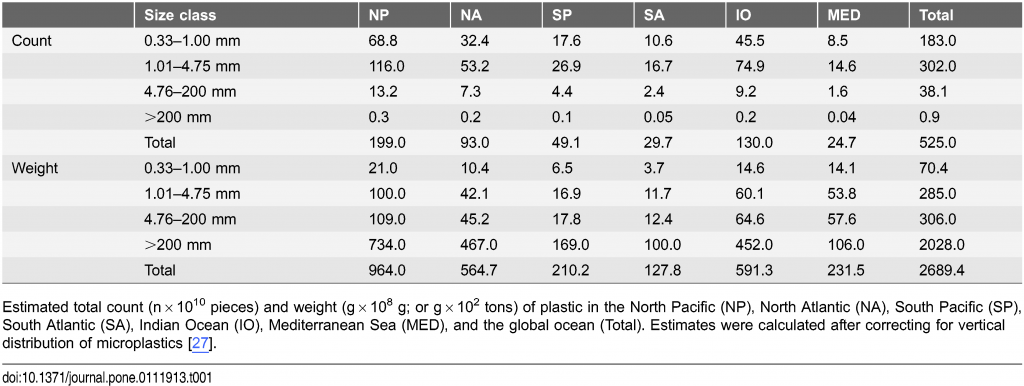
Based on our model results, we estimate that at least 5.25 trillion plastic particles weighing 268,940 tons are currently floating at sea (Table 1). There was a good correspondence between the model prediction and measured data for particle count and weight (Figs. S1 and S2, Table S4). Our estimates suggest that the two Northern Hemisphere ocean regions contain 55.6% of particles and 56.8% of plastic mass compared to the Southern Hemisphere, with the North Pacific containing 37.9% and 35.8% by particle count and mass, respectively. In the Southern Hemisphere the Indian Ocean appears to have a greater particle count and weight than the South Atlantic and South Pacific oceans combined.
Table 1: Model results for the total particle count and weight of plastic floating in the world’s oceans
Of the 680 net tows, 70% yielded density estimates of 1000–100,000 pieces km−2 and 16% resulted in even higher counts of up to 890,000 pieces km−2 found in the Mediterranean. The vast majority of these plastics were small fragments. Although net tow durations varied, the majority of all tows (92.3%) contained plastic, and those locations without plastic were outside the central areas of the subtropical gyres. This pattern is consistent with our model prediction that ocean margins are areas of plastic migration, while subtropical gyres are areas of accumulation. The 891 visual surveys revealed that foamed polystyrene items were the most frequently observed macroplastics (1116 out of 4291 items), while derelict fishing buoys accounted for most (58.3%) of the total macroplastic weight (Table S2). These observations are conservative, recognizing that items with marginal buoyancy, dark color and small size are more difficult to see, especially during challenging environmental conditions (depending on sea state, weather and sun angle).
The data from the four size classes (small microplastics, large microplastics, meso- and macroplastics) were run separately through the model, producing four maps each for count and weight density (Figs. 2 and 3). The mean errors (ε) associated with these predictions can be seen in Table S5. Combining the two microplastic size classes, they account for 92.4% of the global particle count, and when compared to each other, the smallest microplastic category (0.33–1.00 mm) had roughly 40% fewer particles than larger microplastics (1.01–4.75 mm) (Table 1). Most small microplastics were fragments resulting from the breakdown of larger plastic items; therefore we expected the smallest microplastics to be more abundant than larger microplastics. We observed the opposite in all regions globally except in the S. Pacific where large and small microplastic counts were nearly equal.
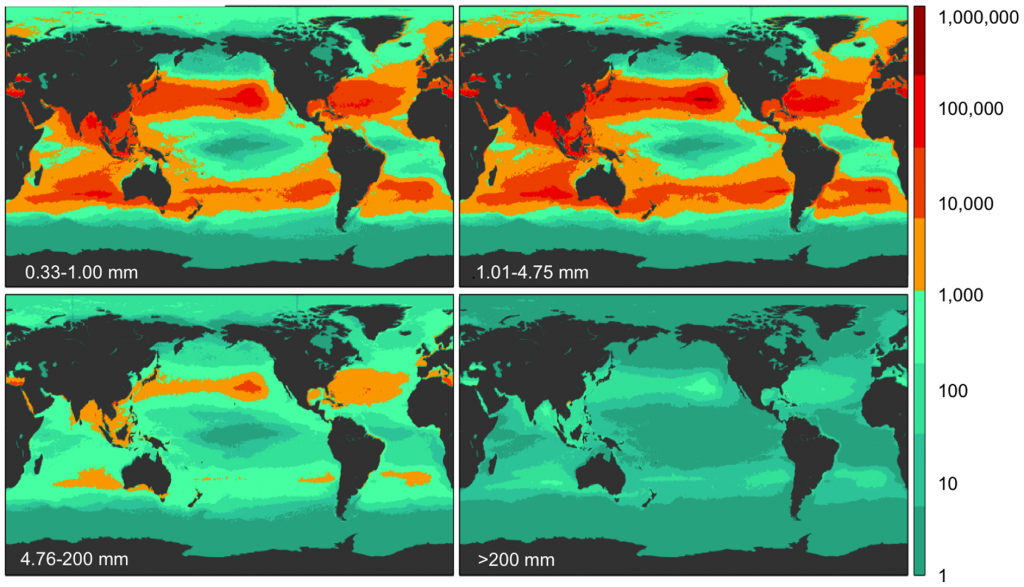
Figure 2. Model results for global count density in four size classes Model prediction of global count density (pieces km−2; see colorbar) for each of four size classes (0.33–1.00 mm, 1.01–4.75 mm, 4.76–200 mm, and >200 mm).
The expected numbers of microplastics (large and small) were an order of magnitude larger than the data-calibrated model counts of microplastics in the world’s oceans (Fig. S3). The expected numbers were derived from conservative estimates of fragmentation from macroplastic to smaller size classes. In contrast to the apparent dearth of microplastics mesoplastics were observed more frequently than expected by the fragmentation ration. For example, in the North Pacific the modeled data show 0.33×1010 particles in the macroplastic size class. Using our estimated fragmentation ratio of 1∶16 between macro and mesoplastic, we expect 5.33×1010 particles in the mesoplastic size class for the entire North Pacific. In this case our modeled data show 13×1010 mesoplastic particles, indicating our fragmentation rates underestimated the data-calibrated model results. This discrepancy could be due to lags in the fragmentation of buoyant mesoplastic and macroplastic, or because mesoplastic items, such as water bottles and single-use packaging, enter the ocean in disproportionate numbers when compared to macroplastic. However, the magnitude of the discrepancy between all size classes suggests that there is differential loss of small microplastics from surface waters.
Figure 3. Model results for global weight density in four size classes Model prediction of global weight density (g km−2; see colorbar) for each of four size classes (0.33–1.00 mm, 1.01–4.75 mm, 4.76–200 mm, and >200 mm). The majority of global weight is from the largest size class.
We found a similar pattern of material loss from the sea surface when comparing the weight of the four size classes. The data showed the weight of plastic pollution globally was estimated to comprise 75.4% macroplastic, 11.4% mesoplastic, and 10.6% and 2.6% in the two microplastic size classes, respectively. Our data suggest that a minimum of 233,400 tons of larger plastic items are afloat in the world’s oceans compared to 35,540 tons of microplastics.
Discussion
This is the first study that compares all sizes of floating plastic in the world’s oceans from the largest items to small microplastics. Plastics of all sizes were found in all ocean regions, converging in accumulation zones in the subtropical gyres, including southern hemisphere gyres where coastal population density is much lower than in the northern hemisphere. While this shows that plastic pollution has spread throughout all the world’s oceans, the comparison of size classes and weight relationships suggests that during fragmentation plastics are lost from the sea surface. Simple comparisons across size classes allowed us to suggest possible pathways for oceanic plastics, and below we discuss these pathways and mechanisms involved.
Plastic pollution is moved throughout the world’s oceans by the prevailing winds and surface currents. This had been shown for the northern hemisphere where long-term surface transport (years) leads to the accumulation of plastic litter in the center of the ocean basins [6], [7]. Our results confirm similar patterns for all southern hemisphere oceans. Surprisingly, the total amounts of plastics determined for the southern hemisphere oceans are within the same range as for the northern hemisphere oceans (Table 1), which is unexpected given that inputs are substantially higher in the northern than in the southern hemisphere [28]. This could mean that plastic pollution is moved more easily between oceanic gyres and between hemispheres than previously assumed [28], leading to redistribution of plastic items through transport via oceanic currents. Furthermore, there might also be important sources of plastic pollution in the southern hemisphere that had not been accounted for, such as currents from the Bay of Bengal that cross the equator south of Indonesia.
Alternatively, a large proportion of plastics might be lost from the sea surface, more so than considered by previous models, and these losses might be disproportionally higher in the northern hemisphere, leading to similar magnitudes in remaining plastic litter at the sea surface. Indeed, stranding of floating plastics on local seashores seems to be more important in the northern than in the southern hemisphere [28], [33]. Other losses (sinking, degradation) may also be responsible for the fact that northern hemisphere oceans contain relative plastic loads that are lower than expected based on global input scenarios. Herein we applied a correction for vertical distribution to all samples related to wind-driven turbulence [27]. Other hydrodynamic processes including downwelling at convergence zones may also influence the vertical distribution of slightly buoyant particles such as microplastics. We suggest that future sampling campaigns use the spatial distribution of sea surface features to better design their sampling efforts and come up with improved global plastic mass inventories.
Other estimates of global and regional weight of microplastic pollution are within the same order of magnitude as our estimates. A study using an 11-year data set in the North Pacific [9] estimates a weight of 21,290 metric tons of floating microplastic, and ours for the same region is 12,100 metric tons. A recent study on the global distribution of microplastic [25] suggests that the total floating microplastic load ranges between 7,000 and 35,000 metric tons, and ours is 35,500 metric tons. This study [25] also found a 100-fold discrepancy between expected microplastic weight and abundance and their observations, indicating a tremendous loss of microplastics. The similarities between our results and those of this study [25] gives us further confidence in our estimates and support our hypothesis that the ultimate fate of buoyant microplastics is not at the ocean surface.
The observations that there is much less microplastic at the sea surface than might be expected suggests that removal processes are at play. These include UV degradation, biodegradation, ingestion by organisms, decreased buoyancy due to fouling organisms, entrainment in settling detritus, and beaching [4]. Fragmentation rates of already brittle microplastics may be very high, rapidly breaking small microplastics further down into ever smaller particles, making them unavailable for our nets (0.33 mm mesh opening). Many recent studies also demonstrate that many more organisms ingest small plastic particles than previously thought, either directly or indirectly, i.e. via their prey organisms [34]–[36]. Numerous species ingest microplastics, and thereby make it available to higher-level predators or may otherwise contribute to the differential removal of small particles from the sea surface, e.g. by packaging microplastics into fecal pellets [37], thus enhancing sinking. Furthermore, there is increasing evidence that some microbes can biodegrade microplastic particles [38]–[40]. This process becomes more important as plastic particles become smaller since at decreasing particle size the surface area∶volume relationship is increased dramatically and oxidation levels are higher, enhancing their biodegradation potential. Thus, bacterial degradation and ingestion of smaller plastic particles by organisms may facilitate their export from the sea surface. In this manner, incorporation of smaller plastics into marine food chains could not only generate impacts on the health of the involved organisms [17]–[20], but also contribute to the removal of small microplastics from the sea surface [37].
Plastics Europe, a trade organization representing plastic producers and manufactures, reported that 288 million tons of plastic were produced worldwide in 2012 [41]. Our estimate of the global weight of plastic pollution on the sea surface, from all size classes combined, is only 0.1% of the world annual production.
However, we stress that our estimates are highly conservative, and may be considered minimum estimates. Our estimates of macroplastic are based on a limited inventory of ocean observations, and would be vastly improved with standardization of methods and more observations. They also do not account for the potentially massive amount of plastic present on shorelines, on the seabed, suspended in the water column, and within organisms. In fact, the larger weight of macroplastic relative to meso- and microplastic, and the global estimate of floating plastic weight relative to the weight of plastic produced annually, indicates that the sea surface is likely not the ultimate sink for plastic pollution. Though significant proportions of meso- and macroplastics may be stranding on coastlines (where some of it could be recovered), removal of microplastics, colonized by biota or mixed with organic debris, becomes economically and ecologically prohibitive, if not completely impractical to recover. This leaves sequestration in sediment the likely resting place for plastic pollution after a myriad of biological impacts along the way, thus reinforcing the need for pre-consumer and post-consumer waste stream solutions to reverse this growing environmental problem.
By generating extensive new data, especially from the Southern Hemisphere, and modeling the plastic load in the world’s oceans in separate size classes, we show that there is tremendous loss of microplastics from the sea surface. The question “Where is all the Plastic?” [42] remains unanswered, highlighting the need to investigate the many processes that play a role in the dynamics of macro-, meso- and microplastics in the world’s oceans.
Figure S1. Comparison of mean and modeled densities. Comparison of data and model predictions for count density (A – pieces km−2) and weight density (B – weight km−2) for four size classes from six ocean regions: North Pacific (NP), North Atlantic (NA), South Pacific (SP), South Atlantic (SA), Indian Ocean (IO), and Mediterranean Sea (MED). https://doi.org/10.1371/journal.pone.0111913.s001
Figure S2. Regression analysis of measured and modeled data. Linear regression of modeled vs. measured values (with correction for vertical distribution) of plastic pollution in terms of count density (A – pieces km−2) and weight density (B – weight km−2) for each of the four size classes. https://doi.org/10.1371/journal.pone.0111913.s002
Figure S3. Comparison of modeled versus expected particle counts (n×1010 pieces) for the global oceans based on conservative fragmentation estimates. The data-calibrated model results of particle count for the global oceans (see Table 1) in each size class differ substantially from conservative estimates of particle counts based on assumed fragmentation of the number if particles in the next-larger size category. We used simple estimates of particle sizes with 0.2 mm thickness and corresponding diameters, and fragmentation factors of 16 for breakdown of a 200 mm diameter particle into particles of 50 mm diameter, 625 for breakdown of a 50 mm diameter particle into particles of 2 mm diameter, and 6.25 for breakdown of a 2 mm particle into particles of 0.8 mm diameter. https://doi.org/10.1371/journal.pone.0111913.s003
Figure S4. Field locations where weight density was measured. Weight density (g km−2) of marine plastic debris measured at 1333 stations from net tows and survey transects for each of the four size classes (0.33–1.00 mm, 1.01–4.75 mm, 4.76–200 mm, and >200 mm). https://doi.org/10.1371/journal.pone.0111913.s004
Table S1. Expeditions contributing field data. 24 expeditions from 2007–13 contributed data collected at 1571 field locations, with count and weight data in four plastic size classes from six regions: North Pacific (NP), North Atlantic (NA), South Pacific (SP), South Atlantic (SA), Indian Ocean (IO), Mediterranean Sea (MED), and circumnaviating Australia (Au. Cirnav.). Locations marked with an asterisk indicate unpublished data and circles show the type of data collected at each expedition. https://doi.org/10.1371/journal.pone.0111913.s005
Table S2. Percent distribution of items from visual survey transects. 4,291 macroplastic items (>200 mm) in nine categories were observed from all visual survey transects conducted in the North Pacific, South Pacific, South Atlantic, Indian Ocean, and Mediterranean Sea. Mean weights for macroplastic items (Extended Data Table 4) were used to determine percent weight distribution. https://doi.org/10.1371/journal.pone.0111913.s006
Table S3. Using beached macroplastic items to determine mean weight. Mean weight of macroplastic items collected from coastal surveys in Chile (eastern S. Pacific), western South Africa (eastern S. Atlantic), east coast United States (western N. Atlantic), and the Hawaiian Islands, was applied to observed macroplastic items drifting in the ocean and then put through the model to calculate global weight densities. The two categories labeled ‘other fishing gear’ and ‘miscellaneous plastics’ were not calculated from weighing items, rather they were assigned a very conservative weight of 10 g. https://doi.org/10.1371/journal.pone.0111913.s007
Table S4. Comparison of measured to modeled means. The measured means of regional count density (pieces km−2) and weight density (g km−2) of plastic in the North Pacific (NP), North Atlantic (NA), South Pacific (SP), South Atlantic (SA), Indian Ocean (IO), Mediterranean Sea (MED), are compared to modeled results. There is generally a good correspondence between the measured and modeled means for each region. https://doi.org/10.1371/journal.pone.0111913.s008
Table S5. Error margins from the linear regression. Average error margin from the linear regression for the count density (pieces km−2) and weight density (g km−2) in the four size classes. https://doi.org/10.1371/journal.pone.0111913.s009
Acknowledgments
We thank the Ocean Research Project for providing microplastic data from the NAG, Diego Miranda and Guillermo Luna-Jorquera for providing the macroplastic data from the SPG, Cat Spina for macroplastic weights from the Hawaiian Islands, and the NOAA Transpacific Marine Debris Survey for macroplastic data from the NPG. The crews and support staff on the expeditions referenced here, specifically the S/V Mir, ORV Alguita, S/V Sea Dragon, and the Stad Amsterdam, were instrumental in sample collection.
Author Contributions
Conceived and designed the experiments: ME LCML HSC MT JCB PGR JR. Performed the experiments: ME LCML HSC MT CJM JCB FG PGR JR. Analyzed the data: ME LCML HSC MT JCB. Contributed reagents/materials/analysis tools: LCML JCB. Wrote the paper: ME LCML HSC MT CJM JCB FG PGR JR. Calculated plastic fragmentation rates: MT. Designed ocean model: LCML JCB. Contributed field data: ME HSC MT CJM FG PGR JR.
References
-
Teuten E, Rowland S, Galloway T, Thompson R (2007) Potential for plastics to transport hydrophobic contaminants. Environ Sci Technol 41:7759–7764. View Article
-
Mato Y, Isobe T, Takada H, Kanehiro H, Ohtake C, et al. (2001) Plastic resin pellets as a transport medium for toxic chemicals in the marine environment. Environ Sci Technol 35:318–324. View Article
-
Rochman C, Browne M, Halpern B, Hentschel B, Hoh E, et al. (2013) Classify plastic waste as hazardous. Nature 494:169–171. View Article
-
Barnes D, Galgani F, Thompson R, Barlaz M (2009) Accumulation and fragmentation of plastic debris in global environments. Philos Trans R Soc Lond B Biol Sci 364:1985–1998. View Article
-
Barnes D, Walters A, Goncalves L (2010) Macroplastics at sea around Antarctica. Mar Environ Res 70:250–252. View Article
-
Law K, Moret-Ferguson S, Maximenko N, Proskurowski G, Peacock E, et al. (2010) Plastic accumulation in the North Atlantic Subtropical Gyre. Science 329:1185–1188. View Article
-
Eriksen M, Maximenko N, Thiel M, Cummins A, Lattin G, et al. (2013) Plastic marine pollution in the South Pacific Subtropical Gyre. Mar Pollut Bull 68:71–76. View Article
-
Goldstein M, Titmus A, Ford M (2013) Scales of spatial heterogeneity of plastic marine debris in the northeast Pacific Ocean,. PloS one 8 View Article
-
Law K, Moret-Ferguson S, Goodwin D, Zettler E, DeForce E, et al. (2014) Distribution of surface plastic debris in the eastern Pacific Ocean from an 11-year dataset. Environ Sci Technol: doi:https://doi.org/10.1021/es4053076.
-
Reisser J, Shaw J, Wilcox C, Hardesty B, Proietti M (2013) Marine plastic pollution in the waters around Australia: Characteristics, concentrations and pathways. PloS one 8 View Article
-
Hinojosa I, Thiel M (2009) Floating marine debris in fjords, gulfs and channels of southern Chile. Mar Pollut Bull 58:341–350. View Article
-
Collignon A, Hecq J, Galgani F, Voisin P, Collard F, et al. (2012) Neustonic microplastic and zooplankton in the North Western Mediterranean Sea. Mar Pollut Bull 64:861–864. View Article
-
Ryan P (2013) A simple technique for counting marine debris at sea reveals steep litter gradients between the Straits of Malacca and the Bay of Bengal. Mar Pollut Bull 69:128–126. View Article
-
Gregory M (2009) Environmental implications of plastic debris in marine settings-entanglement, ingestion, smothering, hangers-on, hitch-hiking and alien invasions. Philos Trans R Soc Lond B Biol Sci 364:2013–2025. View Article
-
Teuten E, Saquing J, Knappe D, Barlaz M, Jonsson S, et al. (2009) Transport and release of chemicals from plastics to the environment and to wildlife. Philos Trans R Soc Lond B Biol Sci 364:2027–2045. View Article
-
Tanaka K, Takada H, Yamashita R, Mizukawa K, Fukuwaka M, et al. (2013) Accumulation of plastic-derived chemicals in tissues of seabirds ingesting marine plastics. Mar Pollut Bull 69:219–222. View Article
-
Bakir A, Rowland S, Thompson R (2014) Enhanced desorption of persistent organic pollutants from microplastics under simulated physiological conditions. Environ Pollut 185:16–23. View Article
-
Wright S, Rowe D, Thompson R, Galloway T (2013) Microplastic ingestion decreases energy reserve in marine worms. Curr Biol 23:1031–1033. View Article
-
Setälä O, Fleming-Lehtinen V, Lehtiniemi M (2014) Ingestion and transfer of microplastics in the planktonic food web. Environ Pollut 185:77–83. View Article
-
Farrell P, Nelson K (2013) Trophic level transfer of microplastic: (Mytilus edulis) to (Carcinus maenas). Environ Pollut 177:1–3. View Article
-
Carson H, Nerheim M, Carroll K, Eriksen M (2013) The plastic-associated microorganisms of the North Pacific Gyre. Mar Pollut Bull 75:126–132. View Article
-
Goldstein M, Carson H, Eriksen M (2014) Relationship of diversity and habitat area in North Pacific plastic-associated rafting communities. Marine Biology Doi:https://doi.org/10.1007/s00227-014-2432-8.
-
Barnes D (2002) Invasions by marine life on plastic debris. Nature 416:808–809. View Article
-
Maximenko M, Hafner J, Niiler P (2012) Pathways of marine debris derived from trajectories of Lagrangian drifters. Mar Pollut Bull 65:51–62. View Article
-
Cozar A, Echevarria F, Gonzales-Gordillo I, Irigoien X, Ubeda B, et al. (2014) Plastic debris in the open ocean. Proc Natl Acad Sci USA doi:https://doi.org/10.1073/pnas.1314705111.
-
Ryan P (2014) Litter survey detects the South Atlantic ‘garbage patch’. Mar Pollut Bull 79:220–224. View Article
-
Kukulka T, Proskurowski G, Morét-Ferguson S, Meyer D, Law K (2012) The effect of wind mixing on the vertical distribution of buoyant plastic debris. Geophys Res Lett 39:1–6. View Article
-
Lebreton L, Greer S, Borrero J (2012) Numerical modeling of floating debris in the world’s oceans. Mar Poll Bull 64:653–661. View Article
-
Cummings J (2005) Operational multivariate ocean data assimilation. Quart J Roy Meteor Soc Part C 131:3583–3604. View Article
-
Black K, Gay S (1990) A numerical scheme for determining trajectories in particle models. In: Bradbury R, editor, Acanthaster and the Coral Reef. A theoretical approach. Springer-Verlag, Berlin. Pp. 151–156.
-
Hidalgo-Ruz V, Gutow L, Thompson R, Thiel M (2012) Microplastics in the marine environment: a review of the methods used for identification and quantification. Environ Sci Technol 46:3060–3075. View Article
-
Smith S (1988) Coefficients for sea surface wind stress, heat flux, and wind profiles as a function of wind speed and temperature. Geophys Res Lett 93:15467–15472. View Article
-
Lumpkin R, Maximenko N, Pazos M (2012) Evaluating where and why drifters die. Journal of Atmospheric and Oceanic Tech 29:300–308. View Article
-
Goldstein M, Goodwyn D (2013) Gooseneck barnacles (Lepas spp.) ingest microplastic debris in the North Pacific Subtropical Gyre. Peer J 184:2–17 View Article
-
Jantz L, Morishige C, Bruland G, Lepczyk C (2013) Ingestion of plastic marine debris by longnose lancetfish (Alepisaurus ferox) in the North Pacific Ocean. Mar Poll Bull 69:97–104 View Article
-
Lusher A, McHugh M, Thompson R (2013) Occurrence of microplastics in the gastrointestinal tract of pelagic and demersal fish from the English Channel. Mar Poll Bull 67:94–99. View Article
-
Cole M (2013) Microplastic ingestion by zooplankton. Environ Sci Technol 47:6646–6655. View Article
-
Zettler E, Mincer T, Amaral-Zettler L (2013) Life in the “plastisphere”: Microbial communities on plastic marine debris. Environ Sci Technol 47:7137–7146. View Article
-
Harshvardhan K, Jha B (2013) Biodegradation of low-density polyethylene by marine bacteria from pelagic waters, Arabian Sea, India. Mar Poll Bull 77:100–106. View Article
-
Balasubramanian V, Natarajan K, Hemambika B, Ramesh N, Sumathi C, et al. (2010) High-density polyethylene (HDPE)-degrading potential bacteria from marine ecosystem of Gulf of Mannar, India. Lett in Appl Microbiol 51:205–211 Plastics Europe (2013) Plastics—the facts 2013: An analysis of European latest plastics production, demand and waste data. Available: www.plasticseurope.de/cust/documentrequest.aspx?DocID=59179 Accessed 2014 Jan 1. View Article
-
Plastics Europe (2013) Plastics—the facts 2013: An analysis of European latest plastics production, demand and waste data. Available: www.plasticseurope.de/cust/documentrequest.aspx?DocID=59179. Accessed 2014 Jan 1.
-
Thompson R, Olsen Y, Mitchell R, Davis A, Rowland S, et al. (2004) Lost at sea: Where is all the plastic? Science 304:838. View Article
1.7 Chapter Summary
1.8 Chapter Homework:
1.9 References:
Chapter 1 materials have been adapted and modified from the following creative commons resources unless otherwise noted:
1. Anonymous. (2012) Introduction to Chemistry: General, Organic, and Biological (V1.0). Published under Creative Commons by-nc-sa 3.0. Available at: http://2012books.lardbucket.org/books/introduction-to-chemistry-general-organic-and-biological/index.html
2. Poulsen, T. (2010) Introduction to Chemistry. Published under Creative Commons by-nc-sa 3.0. Available at: http://openedgroup.org/books/Chemistry.pdf
3. OpenStax (2015) Atoms, Isotopes, Ions, and Molecules: The Building Blocks. OpenStax CNX.Available at: http://cnx.org/contents/be8818d0-2dba-4bf3-859a-737c25fb2c99@12.